Last updated on February 21, 2026 by Editorial Team
Author(s): Saif Ali Kheraj
Originally published on Towards AI.
How agentic AI architectures can compress decision cycles in regulated manufacturing with practical implementation blueprints using CrewAI.
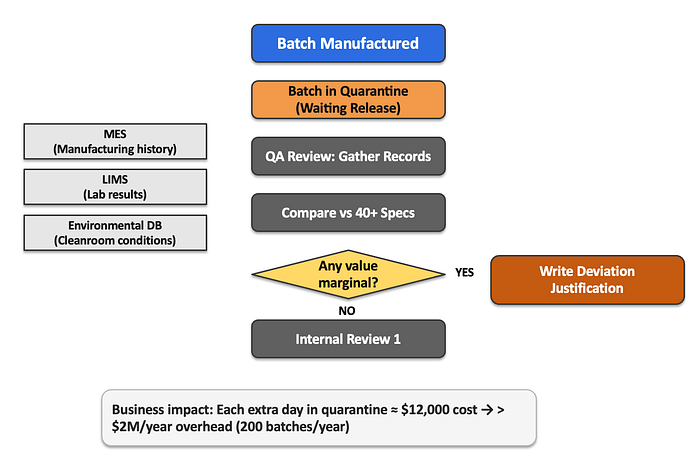
Every contract pharmaceutical manufacturer faces the same operational hurdles. After a drug batch completes production and passes all laboratory testing, it goes into quarantine status, not because there is anything wrong with the drug, but because regulatory review has not yet been completed.
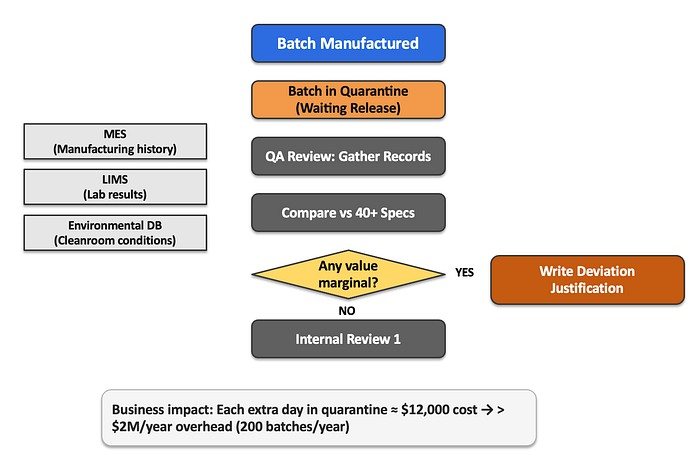
This case study details how XYZ Pharma implemented a multi-agent AI system, reducing the quarantine period for drug batches from 14 days to 3 days while maintaining compliance with regulatory standards. It highlights traditional processes that cause delays, such as the need for careful review of production and laboratory data by quality assurance (QA) teams, and explores the innovative use of AI technologies to streamline decision making and increase operational efficiency in the pharmaceutical manufacturing sector.
Read the entire blog for free on Medium.
Published via Towards AI
We build enterprise-grade AI. We will also teach you how to master it.
15 Engineers. 100,000+ students. The AI Academy side teaches what actually avoids production.
Get started for free – no commitments:
→ 6-Day Agent AI Engineering Email Guide – One Practical Lesson Per Day
→ Agents Architecture Cheatsheet – 3 Years of Architecture Decisions in 6 Pages
Our courses:
→ AI Engineering Certification – 90+ lessons from project selection to deployed product. The most comprehensive practical LLM course.
→ Agent Engineering Course – Hands-on with production agent architectures, memory, routing, and eval frameworks – built from real enterprise engagements.
→ AI for Work – Understand, evaluate, and apply AI to complex work tasks.
Comment: The content of the article represents the views of the contributing authors and not those of AI.